Description
The BCR-ABL Qualitative Test detects the presence of the abnormal “Philadelphia Chromosome.” It is the primary diagnostic tool for Chronic Myeloid Leukemia (CML) and certain types of Acute Lymphoblastic Leukemia (ALL). This test answers a simple “Yes or No”—is the genetic mutation present?
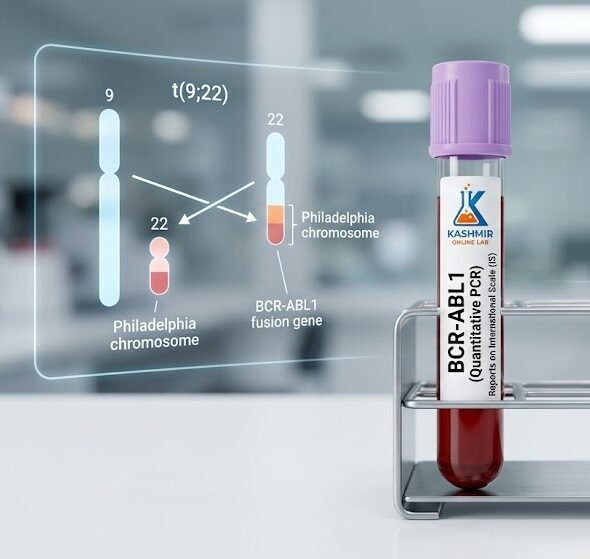
Overview: The BCR-ABL gene is not normal. It is formed when two separate chromosomes (9 and 22) break and accidentally swap pieces—a mutation known as the Philadelphia Chromosome. This new, abnormal gene produces a protein that causes white blood cells to grow out of control, leading to leukemia.
-
Qualitative: This version of the test simply checks for the presence of the gene to confirm a diagnosis.
-
Quantitative: A different version (Quantitative) counts how much of the gene is present to monitor if treatment is working.
Clinical Significance:
-
Diagnosis of CML: It is the hallmark of Chronic Myeloid Leukemia. Almost 95% of CML patients have this mutation.
-
Diagnosis of ALL: It is also found in about 25% of adults with Acute Lymphoblastic Leukemia (ALL).
-
Targeted Therapy: A positive result confirms that the patient is a candidate for “Tyrosine Kinase Inhibitor” (TKI) drugs like Imatinib (Gleevec), which specifically target this protein.
When is this test recommended?
-
Initial Diagnosis: When a Complete Blood Count (CBC) shows very high white blood cell counts and the doctor suspects leukemia .
-
Classification: To distinguish CML from other similar blood disorders.
-
Risk Stratification: In ALL patients, having this gene indicates a need for more aggressive treatment.
Sample Requirements:
-
Specimen: Whole Blood (EDTA) or Bone Marrow.
-
Container: Lavender Top (EDTA) Tube.
-
Preparation: No fasting is required, but the sample must be processed quickly (usually within 24-48 hours) to preserve the RNA.