Description
The BCR-ABL Quantitative Test is the gold-standard molecular test used to diagnose and strictly monitor patients with specific blood cancers, primarily Chronic Myeloid Leukemia (CML). This test measures the exact amount of the abnormal “Philadelphia chromosome” fusion gene in your blood, reported using the mandatory International Scale (IS) to ensure standardization and guide life-saving targeted therapy decisions.
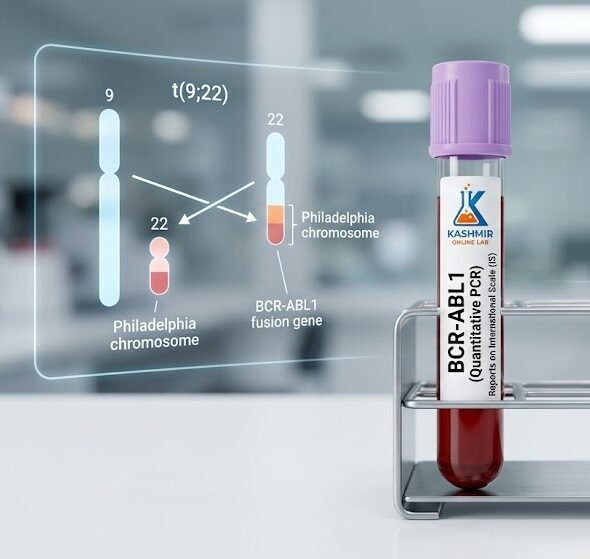
This is a highly advanced molecular genetics test. It looks for the BCR-ABL1 fusion gene, which results from an acquired genetic mutation known as the Philadelphia chromosome. This abnormal chromosome forms when pieces of chromosome 9 and chromosome 22 break off and trade places (t(9;22) translocation). This swap creates a faulty gene that provides instructions to produce an abnormal protein, causing the bone marrow to flood the blood with leukemia cells.
This quantitative test doesn’t just check if the gene is present (qualitative); it measures how many copies of the abnormal gene are active in your cells compared to a normal reference gene.
Clinical Significance: The quantitative BCR-ABL1 test is vital for managing chronic leukemia:
-
Initial Diagnosis Confirmation: While other tests (like FISH) can find the translocation, this test provides a baseline quantity of the disease burden.
-
Monitoring Targeted Therapy (TKI): The standard treatment for CML uses drugs called Tyrosine Kinase Inhibitors (TKIs) that directly attack the abnormal protein made by this gene. This test is the only way to measure how effectively the drug is working.
-
Achieving Major Molecular Response (MMR): A key treatment goal is achieving a Major Molecular Response (MMR), defined as a BCR-ABL1 level of ≤0.1% on the International Scale. This response strongly predicts long-term survival.
-
Detecting Recurrence and Resistance: A rising BCR-ABL1 level in a patient previously in remission can give the first warning sign that the cancer is coming back or that the patient has developed resistance to their current drug, requiring a medication change.
Understanding the International Scale (IS): Because different laboratories can use slightly different methods, the International Scale (IS) was developed to standardize results worldwide. Your lab report will always provide a percentage standardized to this scale. This ensures that a result of 0.1% in Srinagar means the same clinical response as 0.1% in New York, allowing your hematologist to follow established international treatment guidelines.
When is this test recommended?
-
At Diagnosis: To establish a baseline level.
-
Every 3 Months: After starting treatment, patients must be tested every 3 months until they achieve MMR (0.1%IS or lower).
-
Periodically: Once stable, testing frequency may be reduced to every 6 months to monitor for long-term remission.
Sample Requirements:
-
Specimen: Whole Blood (Maternal) is standard for monitoring. Some labs also accept Bone Marrow (BM).
-
Container: Lavender Top (EDTA) Tube.
-
Volume: At least 3 mL to 5 mL of whole blood is required to extract enough high-quality RNA. Minimum 1 mL for children.
-
Preparation: No fasting is required.
-
Crucial Handling: RNA is very unstable. The sample must reach the specialized molecular diagnostics laboratory as soon as possible, ideally within 24–48 hours of collection, to ensure accurate results. Samples cannot be accepted if frozen or clotted.